(c) According to Dalton’s atomic theory of matter, atoms of different elements have different masses. For example, 17Cl 35 and 17Cl 37, both are isotopes. But, it is seen that atoms of same element can have slightly different masses called isotopes. (b) According to Dalton’s atomic theory, all atoms of an element have exactly the same mass. Today we know that an atom can be further divided into smaller particles known as electrons, protons, and neutrons. 
(a) One of the major drawbacks of Dalton’s atomic theory of matter is that atoms had been thought to be indivisible (which cannot be further divided). Drawbacks of Dalton’s Atomic Theory of Matterĭalton’s atomic theory of matter suffered from the following drawbacks or limitations. (b) Atoms of elements take part in the chemical reaction is correct and accepted in modern atomic theory with experimental evidence till today. (a) Dalton’s atomic theory successfully explained the law of conservation of mass and some other laws of chemical combination. There are two main merits of Dalton’s atomic theory. For example, the postulate of Dalton’s atomic theory that “atoms can neither be created nor destroyed nor transformed into atoms of another element in chemical reactions” is the result of law of conservation of mass proposed by Lavoisier.Īnother postulate of Dalton’s atomic theory is that “an element consists of atoms having fixed mass, and relative number and kind of atoms in a given chemical compound is fixed” came from the law of definite proportions proposed by Proust. ---teachoo-01.jpg)
John Dalton proposed his atomic theory based on the laws of chemical combination. (8) When atoms of distinct elements take part in chemical reactions, they involve only reorganisation (i.e. (7) A chemical compound always has the same (or fixed) relative numbers and kind of atoms. Similarly, iron forms several compounds in different ratio, such as FeO (1:1), Fe 2O 3 (2:3), and Fe 3O 4 (3:4). For instance, carbon combines with oxygen to form carbon monoxide in 1:1 proportion and carbon dioxide in 1:2 proportion. (6) Atoms of two elements may combine in different proportions to form more than one compound. 
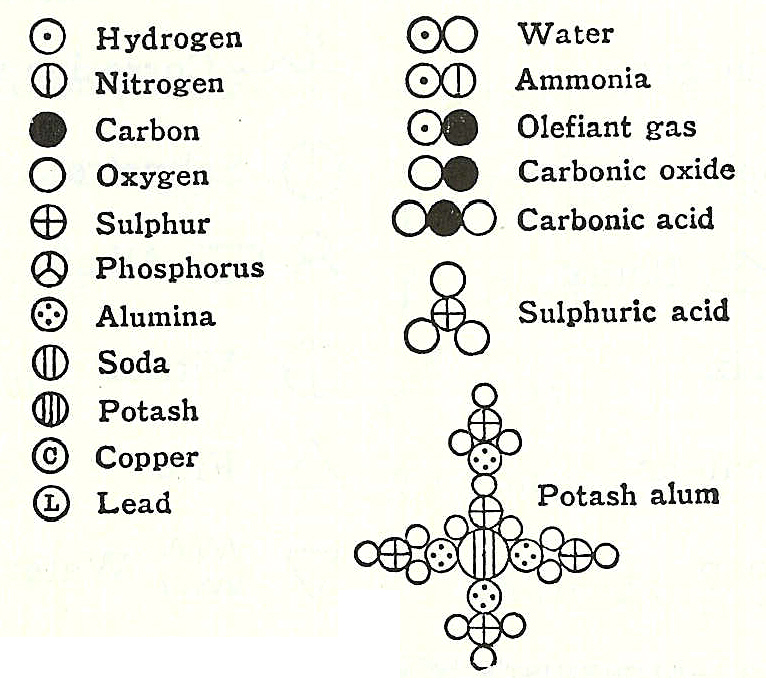
Similarly, calcium combines with chlorine to form CaCl 2 in a fixed ratio 1:2. For example, sodium combines with chlorine to form sodium chloride (NaCl) in a fixed whole number ratio 1:1. (5) Chemical compounds are formed when atoms of different elements combine with each other in a fixed whole number ratios such as 1:1, 1:2, 2:3, and so on. (4) Atoms can neither be created nor destroyed or transformed into atoms of other elements in the chemical reactions. For example, all atoms of Cu are different from atoms of Zn in every respect of their mass, shape, size, and chemical properties. (3) Atoms of different elements are different in mass, size, and chemical properties. Similarly, all atoms of Zn are identical in all respects, such as mass, shape, size, and chemical properties. For example, all atoms of Cu are identical in all respects, such as mass, shape, size, and chemical properties. They have the same mass, shape, size, and chemical properties. (2) All atoms of an element are identical to each other. An atom is the smallest particle of an element that takes part in a chemical reaction. (1) All the matter consists of very small (or tiny) particles called atoms, which are indivisible. The main postulates (or assumptions) of Dalton’s atomic theory of matter are as follows: This atomic theory is mainly based on the laws of chemical combination. This theory is also known as Dalton’s atomic theory of matter. The evidence for atoms is so great that few doubt their existence.In 1808, An English scientist John Dalton proposed the first scientific theory of matter. \): Dalton's symbols from his text "A New System of Chemical Philosophy." 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
